You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
what is this???
- Thread starter ben2363
- Start date

Help Support Prospecting Australia:
This site may earn a commission from merchant affiliate
links, including eBay, Amazon, and others.
Pyrite, tin, antimony, galena..could be many of things, bit tricky to identify a small portion. Poking it with a bit of wood or needle and youll be able to tell if its hard or soft.
The details for the mine may also suggest what it may be. Lots of minerals are common within the goldfields and lots of metals are white/silvery metals too. Most likely a pyrite though :Y:
The details for the mine may also suggest what it may be. Lots of minerals are common within the goldfields and lots of metals are white/silvery metals too. Most likely a pyrite though :Y:
antimony
Defintely not molybdenite and probably too hard for stibnite as well. Arsenopyrite or pyrite. Once you get your eye in the colour is the best clue (pyrite tends to be more brassy in colour, arsenopyrite more silver but they can be similar - I get it right nearly every time but I have seen a lot). Streak and hardness are very similar, both can have parallel striae on their surfaces, neither has good cleavage. Neither dissolves in common, cold acids. They have different crystal forms (you would need a hand lens) - pyrote forms cubes and octahedra but arsenopyrite does not, arsenopyrite twins are common in slate in the Victorian goldfields (in the form of a cross as a rule), pyrite does not and is often seen as disseminated cubes - arsenopyrite crystals never form equidimensional forms (cubes, octahedra) but pyrite forms do. Arsenopyrite crystal fprms tend to be more elongate in one of their dimensions (imagine taking a cube and flattening it by pressing on one of its pointed corners until it becomes elongate).
I will start a blog on mineral identification if there is interest - perhaps both principles of identification and drawing members identification problems together into one place.
I will start a blog on mineral identification if there is interest - perhaps both principles of identification and drawing members identification problems together into one place.
Also not "tin" and probably not galena (tin does not occur as metallic tin but as tin sulphides and oxides that don't have that colour - or a metallic look in the case of the oxide, cassiterite). I would not expect galena to have that "fracture" - it cleaves along planar surfaces even in tiny specimens.
"I will start a blog on mineral identification if there is interest - perhaps both principles of identification and drawing members identification problems together into one place."
One 'yes please'.....I think I've forgotten most of the little I knew!
One 'yes please'.....I think I've forgotten most of the little I knew!
- Joined
- Jan 27, 2016
- Messages
- 274
- Reaction score
- 295
I would be interested in that Goldierocks. I think we all have come across rocks, minerals and gemstones we would like identified but have been unable to determine there exact names. I think any collation of helpful research publications and videos could serve well as a resource that would be a big help for us all in identifying our finds. For example a collection of links that explain the varies physical characteristics of stones and how to test them (what is hardness, how to test it, what is sg and how to determine it etcetera etc). :Y:
silver
silver
- Joined
- Dec 19, 2013
- Messages
- 18,888
- Reaction score
- 25,811
When you first go into forumgoldierocks said:I would if I knew how to start a completely new topic, but I have never been able to find info on that anywhere, and am awaiting a reply to my enquiry to the web admin a short time ago (it is still holidays for many I imagine)
and choose a forum such as Treasure Hunting
and a section such as Treasure Coin and Relic
you will see the start new topic button
and that will take you here (where the magic happens) :Y:
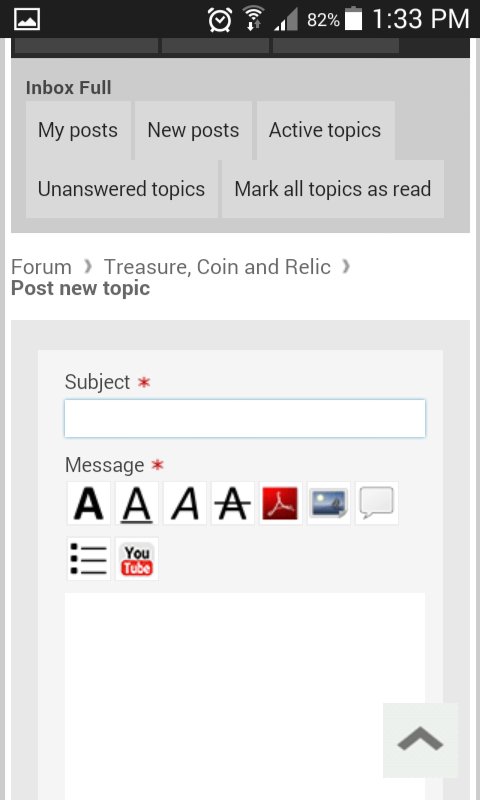
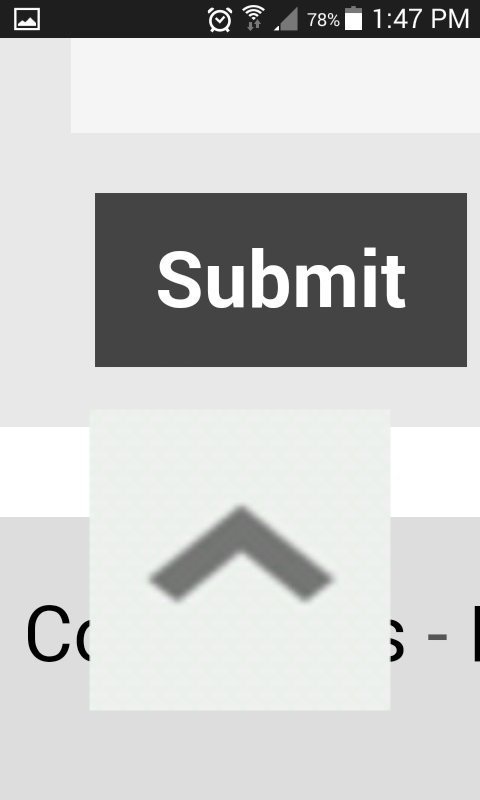
don't forget to hit that submit button at the end though.
:Y:
silver
silver
- Joined
- Dec 19, 2013
- Messages
- 18,888
- Reaction score
- 25,811
No worries mate, someone woulda helped me with that way back when, cause it took me a while as an internet novice to understand all those background functions that have importance to the experience on here.goldierocks said:Thanks Silver - will get onto it after a week or so break.
amazing how we can do fine without something while we don't know about it properly. Great to upskill on here. :Y:
Similar threads
- Replies
- 0
- Views
- 503
- Replies
- 0
- Views
- 137



